PharmaXcelerate USA 2026 Biopharma Manufacturing, Scale-Up & Commercial Readiness
PharmaXcelerate USA 2026 Biopharma Manufacturing, Scale-Up & Commercial Readiness
ABOUT THE CONFERENCE
PharmaXcelerate USA 2026 brings together senior manufacturing leaders, engineers, and decision-makers from the biopharma and biotech industries to address one central challenge: how to successfully scale complex therapies into commercial reality. Taking place on 15–16 September 2026 in Philadelphia, this edition focuses on manufacturing scale-up, facility strategy, and operational readiness.
Across two days, delegates will engage with experts from leading organizations including AstraZeneca, Pfizer, Biogen, Moderna, Takeda, CSL Behring, and Eli Lilly, alongside engineering, quality, and manufacturing leaders shaping the future of biopharma production. The agenda is built around real-world case studies and operational insights, covering critical topics such as multi-modality facility design, Annex 1 compliance and contamination control, scaling biologics and advanced therapies, MS&T transformation, and the integration of AI, digital twins, and data-driven manufacturing systems.
The program is designed to go beyond theory, focusing on what actually determines success at scale. Sessions explore how manufacturers are overcoming bottlenecks in tech transfer, managing multi-site and CMO networks, implementing predictive manufacturing through PAT and data science, and transitioning from reactive compliance to adaptive quality systems.
PharmaXcelerate USA is a focused, senior-level forum for professionals responsible for manufacturing strategy, facility design, process development, and operational performance. Join your peers in Philadelphia to exchange lessons learned, benchmark approaches, and explore the strategies and technologies defining the future of biopharma manufacturing in the US.
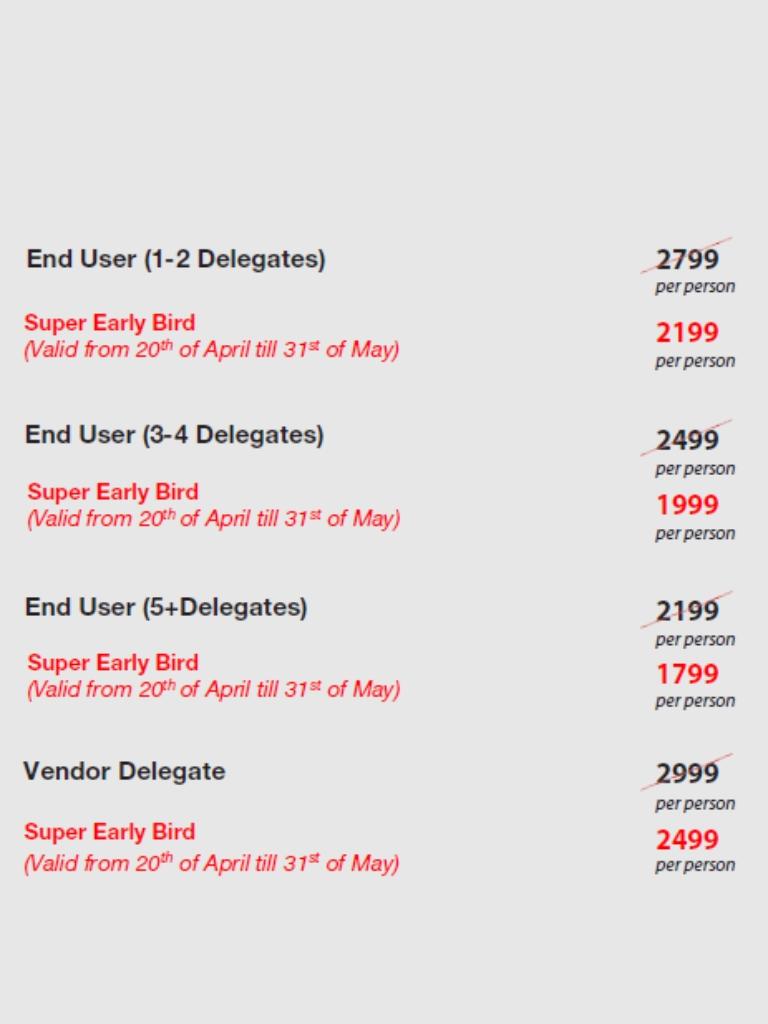
PRICING
Select Currency:
Early Bird Tickets
Purchase Ticket
*Before making your payment, please take a moment to choose your preferred currency. Your convenience matters to us, and selecting the right currency ensures a smooth transaction. Thank you for your attention to this detail!
TOPICS AT A GLANCE:
- Global Biopharma Facility Design for Multi-Modality Manufacturing
- Sustainable Facilities: Annex 1, Energy & Long-Term Performance
- Aligning HSE, Engineering & Manufacturing at Scale
- AI Reshaping Biopharma Quality & Manufacturing
- Predictive Manufacturing via Digital Twin Integration
- PAT & Data Science Pipelines for Real-Time Intelligence
- Global CMC Expectations for mRNA & Advanced Therapies
- Risk & Science-Based Commissioning & Qualification
- Rethinking Quality Frameworks for Advanced Therapies
- Scaling Biologics from Clinical to Commercial Production
SPEAKERS

Edita Botonjic Sehic
Head of Process Analytical Technology & Data Science
Recipharm Advanced Bio

Norman Goldschmidt
Board Member
ISPE

Veera Padmanabhan
Executive Director, Head of Manufacturing Sciences and Technology
AstraZeneca

Aaron Larsen
Chief Scientific Officer,
Quill Therapeutics

Sai Prathyusha Bhamidipati
Director, Reg CMC
Moderna

Hazem Eleskandarani
Senior Director, Global Quality Engineering
CSL Behring

Nick Haycocks
Senior Specialist Quality Assurance
RoNiK

Kim Gauthier
VP, Head of Global Engineering and Facilities
Biogen

Gisèle Fahmi
Director, Acquisitions Collaborations and Divestitures
Pfizer

Mihaela Carmen Simianu
President and Principal Consultant
CONCORDIA MCS

Edwin Harmon
Senior Director, Engineering and Facilities
Eli Lilly

Arjun Channi
Senior Director, Global Regulatory Lead
Takeda

Fouad Atouf
Chief Science Officer
US Pharmacopeia
